Advanced Search
Displaying assets from 1 to 30 out of 14
-
 Description Chemoenzymatic route to sulfoxide 13. a) 3‐(Methylthio)propylamine, HOBt, TEA, EDCI, CH2Cl2; b) meta‐chloroperoxybenzoic acid, CH2Cl2, EtONa, EtOH; c) EtONa, EtOH.
Description Chemoenzymatic route to sulfoxide 13. a) 3‐(Methylthio)propylamine, HOBt, TEA, EDCI, CH2Cl2; b) meta‐chloroperoxybenzoic acid, CH2Cl2, EtONa, EtOH; c) EtONa, EtOH.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Chemoenzymatic synthesis of fluorovinyl dethia motif 11. a) DMPU⋅HF, JohnPhos‐Au‐Pht, dry CHCl3; b) hydrazine monohydrate, EtOH; c) 21, 1‐hydroxy‐1H‐benzotriazole (HOBt), TEA,...
Description Chemoenzymatic synthesis of fluorovinyl dethia motif 11. a) DMPU⋅HF, JohnPhos‐Au‐Pht, dry CHCl3; b) hydrazine monohydrate, EtOH; c) 21, 1‐hydroxy‐1H‐benzotriazole (HOBt), TEA,...
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Chemoenzymatic synthesis of vinyl dethia motif 12. a) Hydrazine monohydrate, EtOH; b) HOBt, TEA, EDCI, CH2Cl2; c) MeONa, MeOH.
Description Chemoenzymatic synthesis of vinyl dethia motif 12. a) Hydrazine monohydrate, EtOH; b) HOBt, TEA, EDCI, CH2Cl2; c) MeONa, MeOH.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Chemoenzymatic synthesis of vinyl thioether 9. a) 1,1′‐dicarbonyldiimidazole, cysteamine⋅HCl, THF, reflux; b) EBX‐TMS, THF, then tetrabutyl ammonium fluoride⋅3 H2O; c) i: AgF, ii: I2, iii: TEA,...
Description Chemoenzymatic synthesis of vinyl thioether 9. a) 1,1′‐dicarbonyldiimidazole, cysteamine⋅HCl, THF, reflux; b) EBX‐TMS, THF, then tetrabutyl ammonium fluoride⋅3 H2O; c) i: AgF, ii: I2, iii: TEA,...
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Chemoenzymatic synthesis of vinyl thioether 10. a) MeONa, MeOH.
Description Chemoenzymatic synthesis of vinyl thioether 10. a) MeONa, MeOH.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
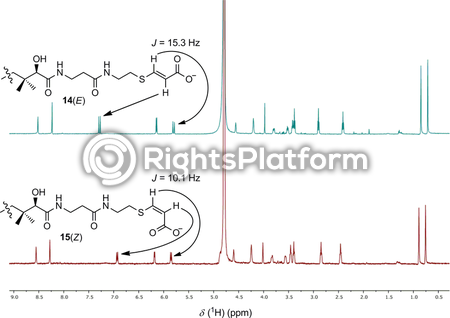 Description 1H NMR spectra of 14(E) and 15(Z); the olefinic resonances of the two geometric isomers are highlighted.
Description 1H NMR spectra of 14(E) and 15(Z); the olefinic resonances of the two geometric isomers are highlighted.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Fluorovinyl cyclohexene 19 is a better inhibitor of the M. tuberculosis dehydroquinase than cyclohexene 20, thus suggesting a fluorine effect in mimicking enolate 18.
Description Fluorovinyl cyclohexene 19 is a better inhibitor of the M. tuberculosis dehydroquinase than cyclohexene 20, thus suggesting a fluorine effect in mimicking enolate 18.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description The citrate synthase reaction progresses through an enolate intermediate 2.
Description The citrate synthase reaction progresses through an enolate intermediate 2.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Historical citrate synthase inhibitors 3–8 drawn relative to intermediate 2.
Description Historical citrate synthase inhibitors 3–8 drawn relative to intermediate 2.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Chemoenzymatic synthesis propenoate thioether isomers 14(E) and 15(Z). a) Methyl propiolate, 1,5‐diazabicyclo[5.4.0]undec‐7‐ene (DBU), THF; b) MeONa, MeOH.
Description Chemoenzymatic synthesis propenoate thioether isomers 14(E) and 15(Z). a) Methyl propiolate, 1,5‐diazabicyclo[5.4.0]undec‐7‐ene (DBU), THF; b) MeONa, MeOH.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description 1H NMR spectra of 9 and 10 clearly showing the difference in the vinyl moiety resonances of these AcCoA enolate analogues.
Description 1H NMR spectra of 9 and 10 clearly showing the difference in the vinyl moiety resonances of these AcCoA enolate analogues.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Steric and electronic organo‐fluorine amide and nucleoside mimetics.
Description Steric and electronic organo‐fluorine amide and nucleoside mimetics.
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Inhibition data for 9–15 with pig heart citrate synthase. AcCoA (50 mm), oxaloacetate (250 mm), Tris buffer (55 mm). The apparent Ki values were determined by using the Cheng–Prusoff equation, as...
Description Inhibition data for 9–15 with pig heart citrate synthase. AcCoA (50 mm), oxaloacetate (250 mm), Tris buffer (55 mm). The apparent Ki values were determined by using the Cheng–Prusoff equation, as...
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem -
 Description Candidate citrate synthase inhibitors 9 to 15(Z).
Description Candidate citrate synthase inhibitors 9 to 15(Z).
Article Title: Acetyl Coenzyme A Analogues as Rationally Designed Inhibitors of Citrate Synthase
Publication Title: ChemBioChem
